1 cubic meter is equal to 1000000 grams, or 1000 liter. The Benefit Of 3-4 Cups Of Tea A Day Is Equal To A Few Liters Of Water, Scientists Say. How To Cook Comparison Table Of Food Weight In Grams - Recipe. of a solute in grams per liter, and see examples that walk through sample problems step-by-step for you to improve your chemistry knowledge and skills. What Nonsense Potato Diet For Daily Weight Loss Of 500 Grams. It uses molar volume of a gas at STP (standard temperature and pressure) This calculator finishes the topic started in Convert moles to liters and liters to moles calculator. Diferent flow rate units conversion from cubic meter per minute to Liters per second. You can view more details on each measurement unit: grams or liter The SI derived unit for volume is the cubic meter. Proper Nutrition Plan For The Week: Kilograms Will Melt Two Liters Of Water Per Day. This online calculator converts grams to liters and liters to grams given a gas formula. Please visit volume flow rate converter for all volume flow units. Type in your own numbers in the form to convert the units Quick conversion chart of grams to liter. Use this page to learn how to convert between grams and liters. Note that rounding errors may occur, so always check the results. Our calculator can also find the mass of substance you need to add to your solution to obtain a desired molar concentration, according to the formula: mass / volume concentration molarity molar mass where mass is the mass of solute (substance) in grams, and volume is the total volume of solution in liters. 
We assume you are converting between gram water and liter. 1 cubic meter is equal to 1000000 grams, or 1000 liter. How many grams in 1 liter The answer is 1000. 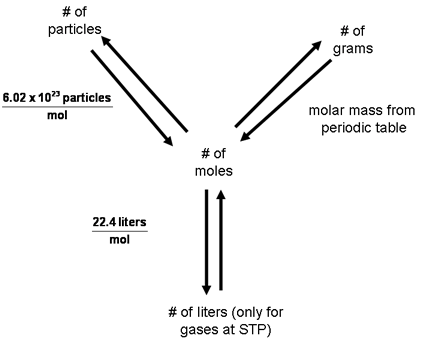
Symbols, abbreviations, or full names for units of length,Īrea, mass, pressure, and other types. More information from the unit converter. You can find metric conversion tables for SI units, as wellĪs English units, currency, and other data. 
The litre is not an SI unit, but (along with units such as hours and days) is listed as one of the "units outside the SI that are accepted for use with the SI." The SI unit of volume is the cubic metre (m³).Ĭonversion calculator for all types of measurement units. The litre (spelled liter in American English and German) is a metric unit of volume. Grams to liters, or enter any two units below: Enter two units to convert From: These calculators give output in fraction of seconds easily.You can do the reverse unit conversion from What mass of oxygen is needed to burn 1 gram of hydrogen, and what mass of water is obtained?įind the relative atomic moss and molecular mass of the each element in the equationĤ grams of hydrogen reacts with 32 grams of oxygen to give 36 grams of water.ġ gram of hydrogen reacts with (32/4) 8 grams of oxygen to give (36/4) 9 grams of water solution.Īt you can see the different chemistry concepts such as pH Calculator, Chemical Reaction Calculator and Chemical Equation Calculator and others. Question: Hydrogen burns in oxygen to form water. Multiply the given moles with molar mass amount of elements with its coefficients to get the weight. This online molarity calculator makes calculating molarity and normality for.0 grams of water ice at a temperature of -20 C to liquid water at the normal. Take the moles of one chemical element to find the weight and mass of products and other elements. Science Chemistry Q&A Library How much energy is needed to boil 45.Calculate the molar mass of each and every reactant and product in the chemical equation.Balance the equation by adding the coefficients to the reactants and products.Follow the simple guidelines mentioned below to solve the Stoichiometry of Chemical Equation. To solve the stoichiometry values we need to balance the equation and predict molar mass. Below you can find the formula for 41g in L if you dont know the density of your ingredient or material make use of our second converter a few lines down. Stoichiometry is used to express the quantitative relationship between reactants and products in the chemical equation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
